In 10 seconds? Engineers have designed an inexpensive battery that could provide cheap backup storage for renewable energy sources, replacing currently used expensive lithium-ion batteries.
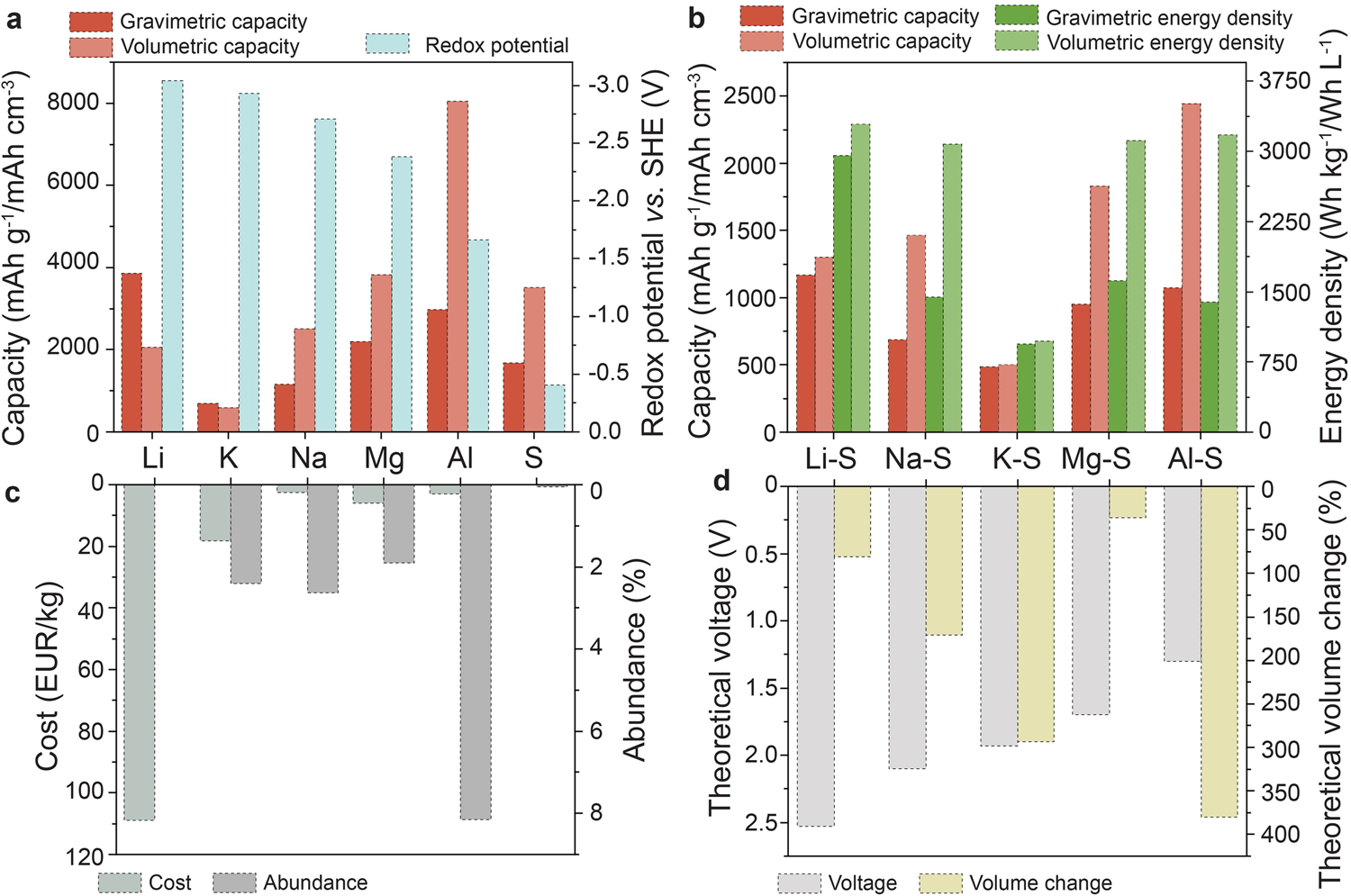
So, what are the alternative materials? This new technology uses inexpensive and abundant materials, aluminium and sulfur, as the two electrode materials with a molten salt electrolyte in between to make batteries that can be used to store energy generated from wind and solar power. These batteries can then be used as power sources at night or on a calm day. The new technology also overcomes one of the biggest problems in battery reliability which is the formation of dendrites.
What are dentrites? Dendrites are spikes of metal that build up on one electrode, rather like icicles and grow towards the other electrode. When this happens the battery short circuits which causes the batteries to fail. However, the molten salt electrolyte used in this new technology prevents this malfunction.
OK, but what’s wrong with the current technology? Lithium-ion batteries are used in our laptops, smartphones, and electric vehicles, as well as for storing energy produced from renewable sources. While lithium-ion batteries have high energy efficiency and can be charged repeatedly, there are several disadvantages which make them less desirable. The two materials used in the batteries, lithium and cobalt, are expensive to mine and can harm both the environment and human health. For example, most of the greenhouse gas emissions that are associated with electric vehicles come from the mining of these metals. In addition, nearly 70% of the cobalt mined around the world comes from the Democratic Republic of Congo (DRC), where child labour is used to mine cobalt and there are serious health and safety concerns.
OK, so how did the researchers decide what metals to use? The researchers wanted to produce a battery that overcame the main issues posed by the lithium-ion variant, such as expense and the flammable material used as the electrolyte. (This makes lithium-ion batteries less than ideal to transport.) The team decided that aluminium was the perfect material as it has the right electrochemical properties for an efficient battery and is also cheap and the most abundant metal on Earth.
And what about the other material? When it came to deciding on the material to use for the second electrode, the researchers looked to use the cheapest non-metal which is sulfur. The final piece in the puzzle was testing what could work as an electrolyte, to carry the ions back and forth between the electrodes during charging and discharging. They trialed several polymers before using a molten salt solution. This solution is ideal as it has a low melting point which means that it doesn’t require special insulation like other batteries. Combined, the ingredients also make the batteries much safer than other options as they cannot burn.
Great, so what can these batteries be used for? The application of this new technology could be far-reaching, from powering a single home or small to medium-sized business to being used in electrical vehicle charging stations. This could help overcome current issues with electrical vehicle (EV) charging infrastructures such as slow charging or the lack of charging infrastructure which are major obstacles in the use of EVs.
So, how can they make the switch to EVs easier? These batteries could be the solution to improving electric charging infrastructure by creating high-power charging stations that could cope with an increase in charging demand and provide charging speeds comparable to filling your car at the petrol pump. These batteries could potentially help overcome the limitations of lithium batteries while costing one-sixth of the cost.
Exciting! What are the next steps? The patents for this system have already been licensed by a company called Avanti, which is a spin-off company co-founded by some of the researchers involved in this project (So, yes – some of the authors have a financial interest). But before you become a full-on skeptic: spin-off companies are a means of technology transfer and can help commercialize academic research, developing it for use in society. Moving forward, the business will need to examine if this new technology can work on a larger scale which will involve subjecting it to a range of stress tests and running it through hundreds of charging cycles to ensure that it meets all the requirements. If successful, this new technology could help in the fight for decarbonisation and provide a solution of adequate storage for renewable energy sources.
You may not know this, but lithium-ion batteries are everywhere ….
… and are used to power our laptops, e-bikes, smartphones, electric vehicles, electric toothbrushes, and solar power backup storage.
They hold a record for the world’s smallest and largest battery! The record for the smallest battery in the world is a lithium-based rechargeable battery developed by researchers at Rice University which is over 60,000 times smaller than an AAA battery and is used to run really small electronic devices.
The largest battery is a giant Tesla battery found in South Australia. This battery is hooked up to a wind power plant and can be used to provide power to more than 30,000 homes!
Lithium batteries are seen as the future for storing renewable energy and shifting to a more climate-friendly planet, but could we be in danger of running out? The International Energy Agency (IEA) predicts we could face shortages as early as 2025! Making discoveries like this one is even more important!
Dr. Lindy Whitehouse has distilled 8 research papers saving you 28 hours of reading time.

The Science Integrity Check of this 3-min Science Digest was performed by Dr. ASM Mainul Hasan.






